In our research we are interested in selective transformations and aim to merge concepts from biocatalysis with semiconductors. Spanning from regio-, chemo-, to enantioselectivity all being achieved by metal-free or first-row transition metal catalysis combined with electrosynthesis and photoredox catalysis.
Here are some glims into our research:
We aim to synthesize (non-canonical) amino acids (ncAA) like various cyclic or acyclic, tertiary or quarternary, di- or oligo amino acids for a diverse set of applications ranging from potential drug molecules to materials with advanced properties.
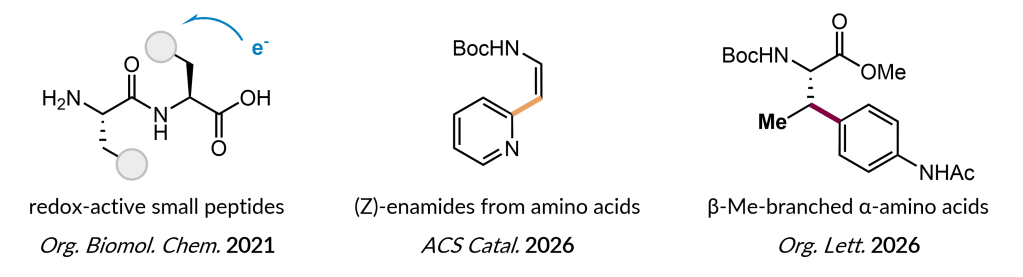
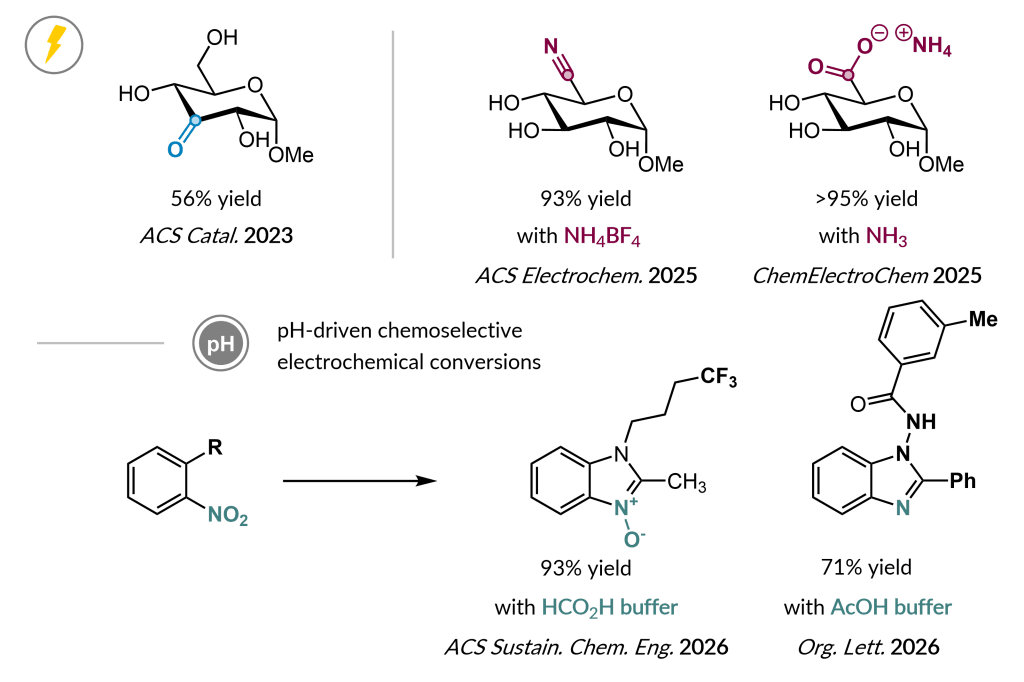
Based on long-standing expertise, another research area continues in electrochemical oxidation and reduction reactions and catalysis. Mild reaction conditions for site-selective oxidations are developed to obtain rare sugars and value-added heterocycles. We are particularly interested in the pH as tunable parameter for chemoselectivity.
Moreover, we are interested in more sustainable chemical reactions. Hence, we discovered organic carbonates, which allow for tunable conversions, thus replacing more harmful solvents (Green Chemistry). Likewise, halogenated organic pollutants must be degraded, but ideally their halides are refurbished into the chemical value chain (Chem). Unsatisfactory reproducibility hampers the acceptance of chemical methods and therefor its application in commercial strategies. These challenges must be addressed across the interdisciplinary scientific communittee (JACS Au).
Our Synthetic Tools
Visible light photo(redox) catalysis merged with abundant transition metals enables powerful transformations under mild conditions. Our group is interested in employing this technique to improve otherwise challenging and long-sequence syntheses.

Redoxchemistry can be purely fueled by electrons via electrochemical setups. In our group we develop innovative electroorganic processes and syntheses to obtain value-added materials.

Our Funding Sources



